Investigate the structure and function of the meiotic centromere and kinetochore
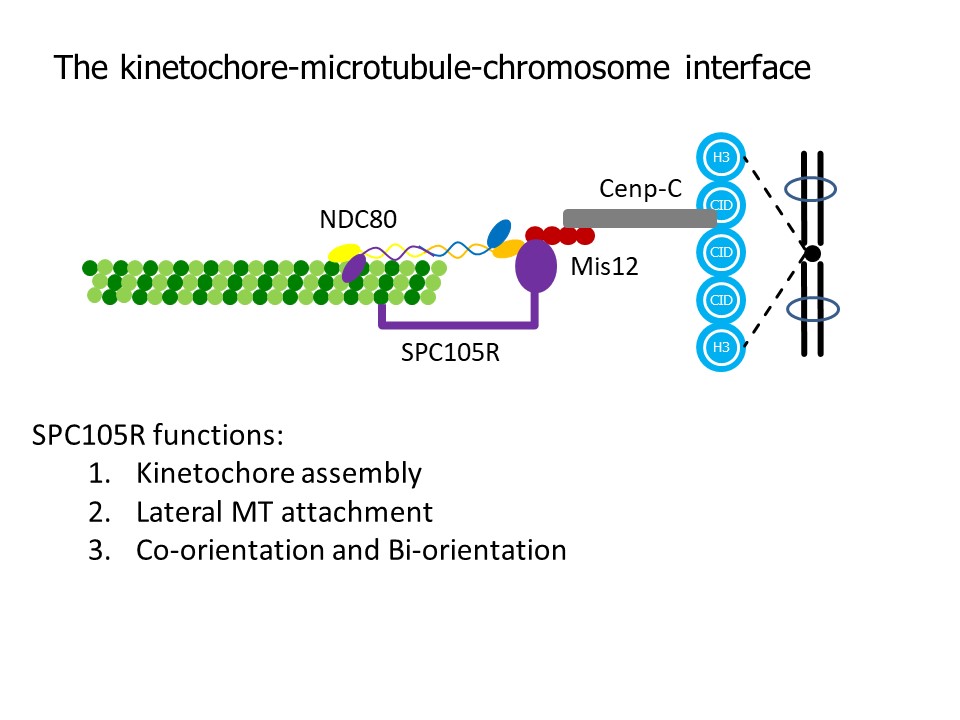
The conserved KMN network is required for KT-MT attachments in vivo and is composed of three groups of proteins: SPC105/KNL1, the MIS12 complex and the NDC80 complex. NDC80 is required for end-on MT attachments, which are important for moving the chromosomes towards the poles at anaphase. We have identified several activities that depend on SPC105R, kinetochore assembly (NDC80 recruitment), lateral microtubule attachments, and sister centromere fusion. The latter is important for coorientation, the mechanism that promotes attachment of sister kinetochores in meiosis I to microtubules from the same pole.
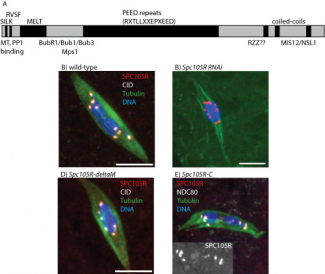
To study the function of SPC105R, we use tissue-specific RNAi to deplete the protein in oocytes. To develop a system to study the functions of SPC105R in oocytes with mutants, we generated an Spc105R RNAi-resistant transgene. This transgene rescues all Spc105R RNAi phenotypes. Using this construct, we are generating RNAi-resistant transgenes with deletions of each SPC105R domain proposed to recruit proteins with specific functions (Figure 1). We are particularly interested in separation-of-function mutants that identify the region(s) required for lateral attachments and sister centromere fusion. Surprisingly, the large central domain and the N-terminal MT-binding domains are not essential for meiosis. However, a transgene expressing only the C-terminal domain appears to be sufficient to localize to the centromeres, recruit other kinetochore proteins, and most surprising, recruits cohesin protection proteins such as MEI-S332/Shugoshin. These studies are continuing with the generation and characterization of additional mutations.
Page Components