Characterize the relationship between the CPC and phosphatases
Prior to meiosis, the DNA replicates. Cohesin is the protein complex that holds these sister chromatids together until meiosis I and II. Cohesin regulation is complex because it must be protected from destruction prior to these stages, and then released in stages. First the chromosomes arms during meiosis I, and then the centromeres and the pericentromeric regions in meiosis II. regulate sister centromere fusion. The analysis of kinetochore proteins SPC105R/KNL1 and Protein Phosphatase 1 (PP1-87B) has shown that two independent mechanisms maintain sister centromere cohesion in Drosophila oocytes. Maintenance of sister centromere cohesion by SPC105R involves protection against Separase, the enzyme that’s destroys cohesin proteins. In contrast, maintenance of sister centromere cohesion by PP1-87B does not involve Separase. Instead, PP1-87B maintains sister centromere cohesion by inhibiting stable KT-MT attachments. In a recent paper describing this work, we propose two mechanisms regulate co-orientation in Drosophila oocytes. First, SPC105R protects cohesins at sister centromeres to maintain fusion. Second, separation of sister centromeres in meiosis II occurs without degrading cohesins, depends on end-on microtubule attachments, and during meiosis I is prevented by PP1-87B mediated destabilization of these attachments.
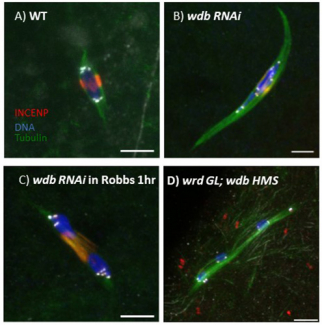
SPC105R maintains sister centromere cohesion during meiosis I by recruiting Protein Phosphatase 2A (PP2A), which modifies cohesins so they cannot be destroyed. What regulates stepwise removal of cohesion in the pericentromeric regions and the chromosome arms is not kown. This analysis is complicated by the fact that Drosophila have two PP2A subunits, WDB and WRD, that are partially redundant. In oocytes lacking SPC105R, WDB does not localize to the centromeres. Wdb RNAi oocytes, however, show a mild defect in cohesion, displaying occasionally showing separated centromeres and precocious anaphase. When both WDB and WRD are depleted by RNAi, we observed a complete failure to maintain sister chromatid and arm cohesion in metaphase I oocytes (Figure 2). We are currently investigating how PP2A is recruited to the pericentromeric regions and chromosome arms. Based on published work in mitotic cells, we are determining if Dalmatian, the Drosophila homolog of cohesin regulator Sororin, recruits PP2A to the chromosomes in addition to MEI-S332/Shugoshin. Our preliminary data shows that Dalmatian is localized to the meiotic chromosomes during early prophase. It may protect cohesion during the long prophase arrest experienced by oocytes, and during meiosis I may be redundant with MEI-S332/Shugoshin.